Alternative Two Step Purification of a Soluble Receptor Fc Fusion Biologic Using Mixed-Mode Chromatography
Principal Investigator Heidi Jones Bio-Rad
Principal ScientistScott Conrad McCarthy PI Celkem
Abstract
This study examines the purification of a biologic fusion protein, EGFR:Fc, employing non-affinity mixed-mode chromatography methods. The performance of these approaches is compared with conventional protein A affinity purification, emphasizing practical considerations within biologic protein purification workflows. An EGFR:Fc fusion protein, expressed in HEK 293 cells, was captured and polished using Bio-Rad’s mixed-mode resins Nuvia™ wPrime2A and Nuvia™ HPQ. Initial capture on Nuvia™ wPrime2A at pH 6.8 resulted in approximately 90% purity, although minor product-related impurities remained detectable. Further purification on Nuvia™ HPQ enhanced purity to over 98%, achieving levels comparable to those obtained via Protein A affinity purification. The two-step non-affinity purification process yielded 70–80% of EGFR:Fc, whereas protein A affinity purification produced yields exceeded 90%. Both purification strategies delivered biologics with analogous binding characteristics and impurity profiles. This investigation offers valuable insights into the application of mixed-mode chromatography for biologic protein purification.
Introduction
Biologics frequently harness key metabolic proteins, such as extracellular domain receptors, to trigger specific biological actions. A notable example is AMGEN’s Enbrel™, a soluble receptor engineered with an IgG Fc domain to enhance half-life and molecular capture. In our study, we explored the purification of a soluble EGFR receptor fused to an Fc domain, utilizing mixed-modal chromatography media. This approach demonstrated results comparable to traditional affinity purification. While protein A-based media is commonly employed, its low pH elution can induce aggregation or chemical modification of fusion biologics. Mixed-modal chromatography offers a neutral alternative, potentially mitigating these issues.
Fig. 1 Mab Select Sure ™ Protein A Chromatogrpahy (Cytiva)
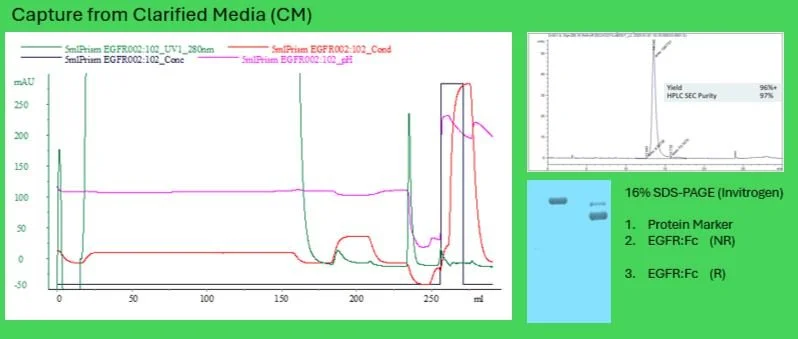
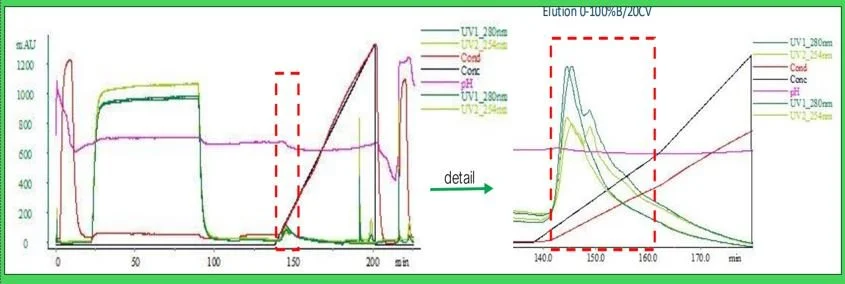
Fig. 2 Capture of CM on Nuvia wPrime 2A Mixed Modal Media
The EGFR:Fc fusion protein was successfully captured on Nuvia™ wPrime2A in 20 mM NaPO₄ (pH 6.8), with this buffer concentration providing enhanced pH stability. Elution fractions were analyzed by reducing gel electrophoresis, revealing a capture purity of 90%.
Polishing on Mixed Modal Chromatography (MCC)
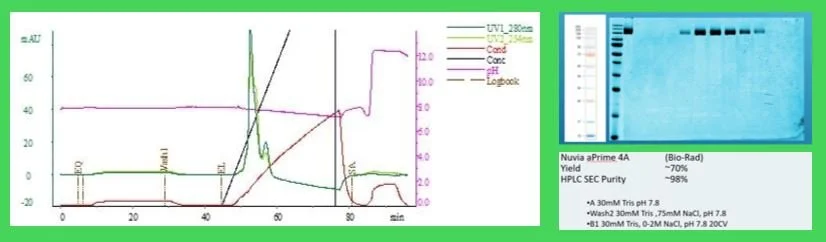
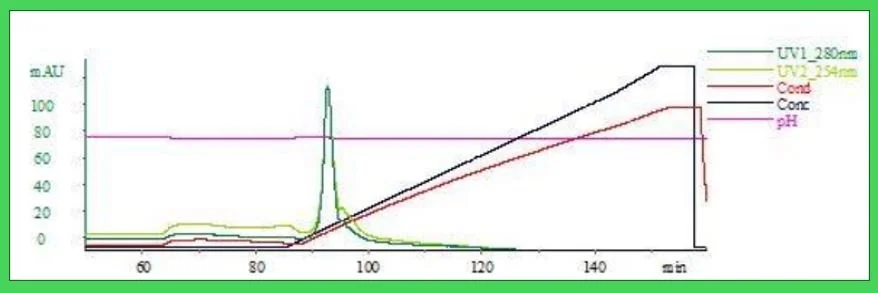
Fig. 3 Polishing on Nuvia aPrime 4a post Nuvia wPrime 2a. The aggregate was reduced below 2% in 30m.M Tris, pH 7.8 with a 75mM wash step. Yields post wPrime yielded ~70% product and ~83% post protein A. Note that there is a shoulder peak which was determined to be non-proteinatious, possible chromaphore from the cell media.
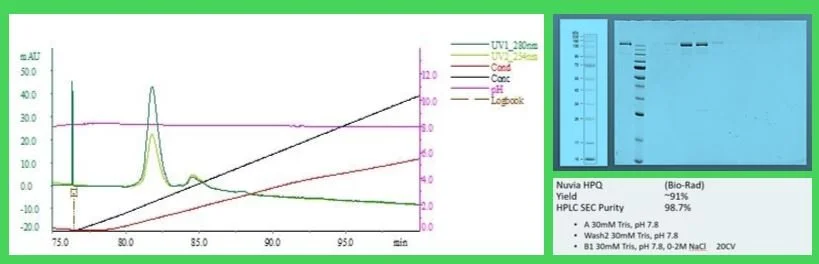
Fig. 4 Nuvia HPQ post wPrime Capture. Nuvia aPrime elution dil 1:5 in buffer A; Conductivity below 10mS.
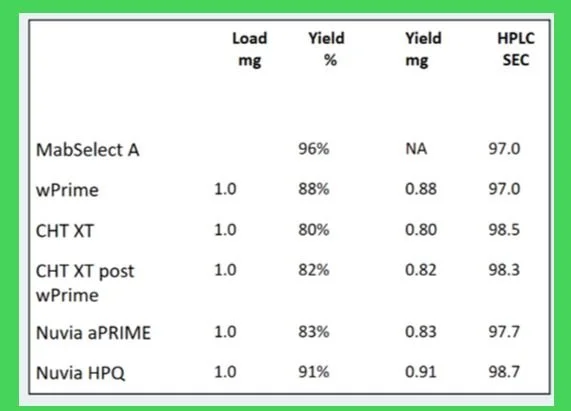
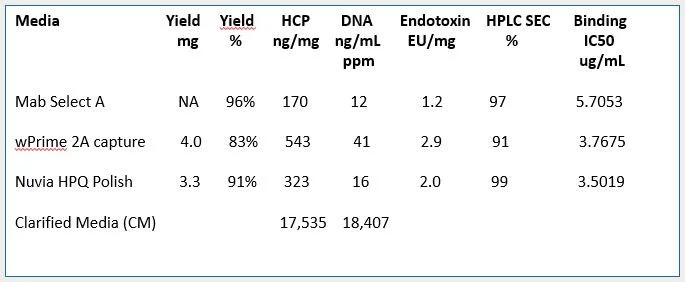
Table 1 Results of Polishing post protein A or wPrime Capture on a 1mL Forsight ™ Column. The condition which met our immediate interests was the HPQ media.
End to End Purification
Fig. 5 The EGFR:Fc fusion protein was successfully captured on Nuvia™ wPrime2A in 20 mM NaPO₄ (pH 6.8), with this buffer concentration providing enhanced pH stability. Elution fractions were analyzed by reducing gel electrophoresis, revealing a capture purity of 90%—notable for a non-affinity medium. Additional bands at 34 and 28 kDa were observed, with Western analysis used to determine their relation to the product. Residual DNA and host cell protein (HCP) levels were also assessed.
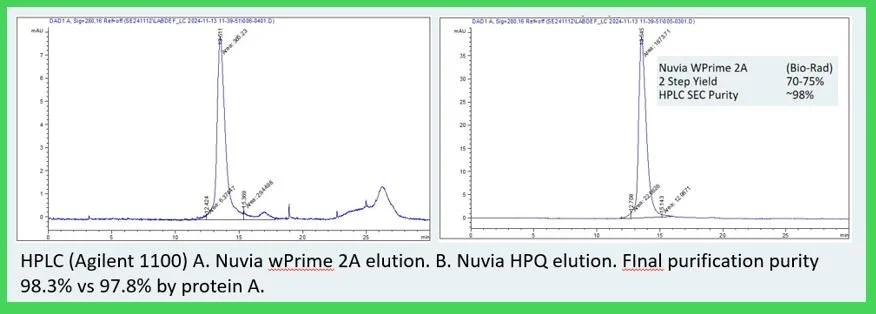
Fig. 6 Polishing on Nuvia™ HPQ resulted in purity exceeding 98% by HPLC SEC. Comparative SDS-PAGE analysis showed similar purity to Protein A-purified Fc fusion protein. Host cell protein was quantified using HEK CHP ELISA, and product activity was confirmed via Cetuximab™ biosimilar binding in an ELISA format.
Fig. 7 HPLC SEC of the Captured EGFR:Fc and Polished Product on HPQ (Bio-Rad)
Table 2 Overall yield was over 70%. Final aggregate was below 1% by HPLC SEC. Clearance of HCP protein and residual DNA were comparable to Protein A.. Binding of the Fc fusion protein was retained against the common biologic antibody Cetuximab.
Primary References
Bulletin 7112 Bio-Rad
Bulletin 7142 Bio-Rad
Bulletin 7080 Bio-Rad
Bio-Rad and Nuvia(™) are trademarks of Bio-Rad Laboratories Inc. in certain jurisdictions. All trademarks used herein are the property of their respective owner.
Acknowledgements
Heidi Jones, Chris Weinert, Elham Ettehadieh, Jenna McKee-Johnson, Bio-Rad, and RAIN Incubator